President Director of Bio Farma, Honesti Basyir, said that the basis for selecting the COVID-19 vaccine must meet several factors such as meeting the elements of safety, effectiveness and quality. This is what determines the government choosing the Sinovac vaccine. In addition, it must be able to prove it from trials starting from preclinical testing, clinical trials in phase one, two and three.
“If you look at the time or development process, the COVID-19 vaccine candidate from Sinovac is one of the 10 fastest candidates and has entered the stage 3 clinical trial. In addition, Bio Farma has mastered the method of making vaccines using an inactivated virus platform,” Honesti said in a written statement, Friday (11/12/2020).
Furthermore, the truth is in terms of the method of making vaccines using an inactive platform or a virus that has been killed and has been proven in other types of vaccines. “The platform or method has been mastered by Bio Farma,” he said.
Then another determining factor is the quality system owned by Sinovac which has been recognized by the World Health Organization (WHO). In the collaboration between Bio Farma and Sinovac, there is also transfer technology in the necessary testing tests.
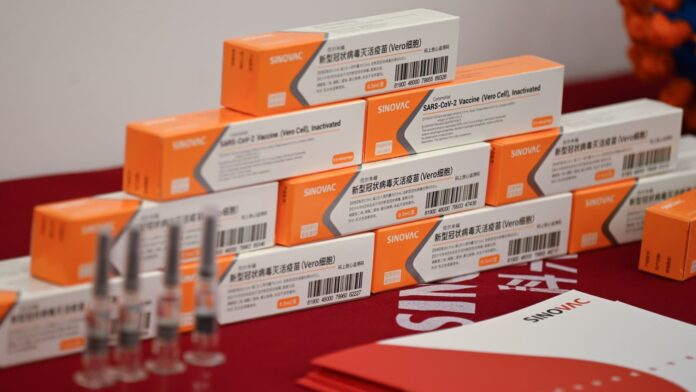
As is known, on Sunday night (6/12/20), as many as 1.2 million doses of the Sinovac COVID-19 vaccine made in China arrived in Indonesia. The total amount of Sinovac vaccine that Bio Farma will bring in is 3 million doses.
After 1.2 million doses arrived on December 6, 1.8 million doses will arrive in late December or early January 2021. In addition to the finished vaccine, in December 2020, Bio Farma also received 15 million doses of vaccine raw materials. Then an additional 30 million more vaccine raw materials will arrive in January 2021.
The vaccine is packaged in seven special Envirotainer containers for quality service during the flight from Beijing to Jakarta. On arrival at Soekarno-Hatta International Airport, the vaccines were taken to Bio Farma, Bandung for quality control and distribution. After obtaining the use permit and halal certification, the vaccination process will be carried out.